HER2-positive Breast Cancer Neoadjuvant Regimens
What neoadjuvant regimens are available for HER2-positive, HR-negative breast cancer?
Based on the retrieved 2024 CSCO Breast Cancer guidelines, the standard neoadjuvant approach for HER2-positive breast cancer is centered on anti-HER2 targeted therapy combined with chemotherapy. The HR-negative status does not alter the core HER2-directed strategy.
### Neoadjuvant Regimens for HER2-Positive, HR-Negative Breast Cancer
The primary recommendation is a dual-targeted regimen, with single-agent trastuzumab as an alternative.
| Regimen | Components | Evidence & Recommendation | Key Notes |
|---|---|---|---|
| **THP (Dual-Targeted)** | Taxane (T) + Trastuzumab (H) + Pertuzumab (P) | **Recommended option**. Supported by the NeoSphere trial (increased pCR rate vs. TH) and the PEONY trial (confirmed efficacy/safety in Asian populations)[1]. | This is the preferred strategy for patients qualifying for neoadjuvant therapy. The guideline notes that in the PEONY trial, surgery was performed after 4 cycles, which is a consideration for clinical practice[1]. |
| **TH (Single-Targeted)** | Taxane (T) + Trastuzumab (H) | **Standard agent**. Clinical studies have demonstrated that this combination significantly increases the pCR rate[1]. | This remains a valid option. The expert group generally acknowledges that dual-targeted therapy (THP) may be considered for all patients who qualify for single-targeted therapy[1]. |
### Clinical Decision Summary
- **First-Line Consideration**: The **THP regimen** (Taxane + Trastuzumab + Pertuzumab) is the guideline-supported, evidence-based choice for maximizing pCR rates in HER2-positive disease, regardless of HR status.
- **Alternative**: The **TH regimen** (Taxane + Trastuzumab) is a standard option, particularly if dual-targeted therapy is not feasible due to patient factors, tolerability, or access.
The HR-negative status does not change the selection of these HER2-directed backbones. The choice between THP and TH should be guided by patient eligibility, risk-benefit assessment, and the goal of achieving a pathological complete response (pCR).
---
### Further Diagnostic Suggestions (Reference Only)
To enable a more accurate assessment, if available, please consider providing:
* **Tumor Stage (T/N)**: The guideline specifies neoadjuvant therapy for tumors >2 cm, positive lymph nodes, or inflammatory breast cancer. Staging confirms the indication.
* **Patient's Cardiac Function (LVEF)**: Baseline LVEF assessment is critical before initiating trastuzumab and pertuzumab, as they carry a risk of cardiotoxicity.
---
*Disclaimer: This response is AI-generated based on the retrieved literature (CSCO 2024 guidelines) and is intended for clinical reference only. Please verify the full guideline text and apply individual patient judgment in clinical decision-making.*
HER2-positive HR-negative breast cancer neoadjuvant pCR prognostic value
## Prognostic Value of Pathologic Complete Response (pCR) in HER2-Positive, HR-Negative Breast Cancer
Achieving a pathologic complete response (pCR) following neoadjuvant therapy in HER2-positive, HR-negative breast cancer carries **strong prognostic significance**, though the strength of this association varies by subtype.
### Key Evidence Summary
| Aspect | Finding | Source |
|--------|---------|--------|
| **pCR Definition** | Absence of any residual invasive cancer on H&E evaluation of resected breast specimen and all sampled ipsilateral lymph nodes (ypT0/is ypN0) | Regulatory definition [7] |
| **Prognostic Strength by Subtype** | Correlation between pCR and long-term outcomes is **strongest for triple-negative**, **less so for HER2-positive**, and **least for HR-positive disease** | NCCN 2025 [2] |
| **pCR as Surrogate Endpoint** | pCR is supported by regulatory agencies as a surrogate endpoint for accelerated approval; however, association is proven at **patient level** (pCR achievers have better survival) but **not consistently at trial level** (poor association between pCR rate improvement and survival benefit across trials) | [11] |
| **Residual Disease Impact** | Residual invasive disease is the **most significant adverse prognostic factor** for DFS and OS in HER2+ breast cancer | Canadian Consensus [5] |
| **pCR Rates in HER2+ Disease** | Higher pCR rates are observed in HER2-positive and triple-negative subtypes compared to HR-positive/HER2-negative disease | UEMS Guide [10] |
### Clinical Implications
**For patients who achieve pCR:**
- Significantly improved long-term disease-free survival (DFS) compared to those with residual disease [5]
- Favorable prognosis, though the correlation is less robust than in triple-negative breast cancer [2]
**For patients with residual disease:**
- Represents the most significant adverse prognostic factor [5]
- Guides escalation of adjuvant therapy (e.g., T-DM1 per KATHERINE trial paradigm) [4]
- Residual cancer burden (RCB) or Neo-Bioscore may further refine prognostic stratification [10]
### Important Caveats
1. **Trial-level surrogacy limitation**: While pCR is a meaningful clinical endpoint at the individual patient level, improvements in pCR rates across trials do not consistently translate to proportional survival benefits [11]
2. **Dichotomous limitation**: pCR as a binary endpoint ignores that partial responses are associated with better outcomes than no response; RCB classification provides more granular prognostic discrimination [10]
3. **Receptor conversion**: Following neoadjuvant therapy, HER2 loss is associated with significantly poorer DFS (HR 1.92, 95% CI 1.51–2.43; p<0.001) and OS (HR 2.20, 95% CI 1.44–3.84; p<0.001), underscoring the importance of reassessing receptor status on residual disease [4]
### Guideline Recommendations on Post-Neoadjuvant Management
The following flowchart illustrates the standardized treatment pathways for HER2-positive early breast cancer based on clinical staging, surgical outcomes, and pathological response to neoadjuvant therapy:
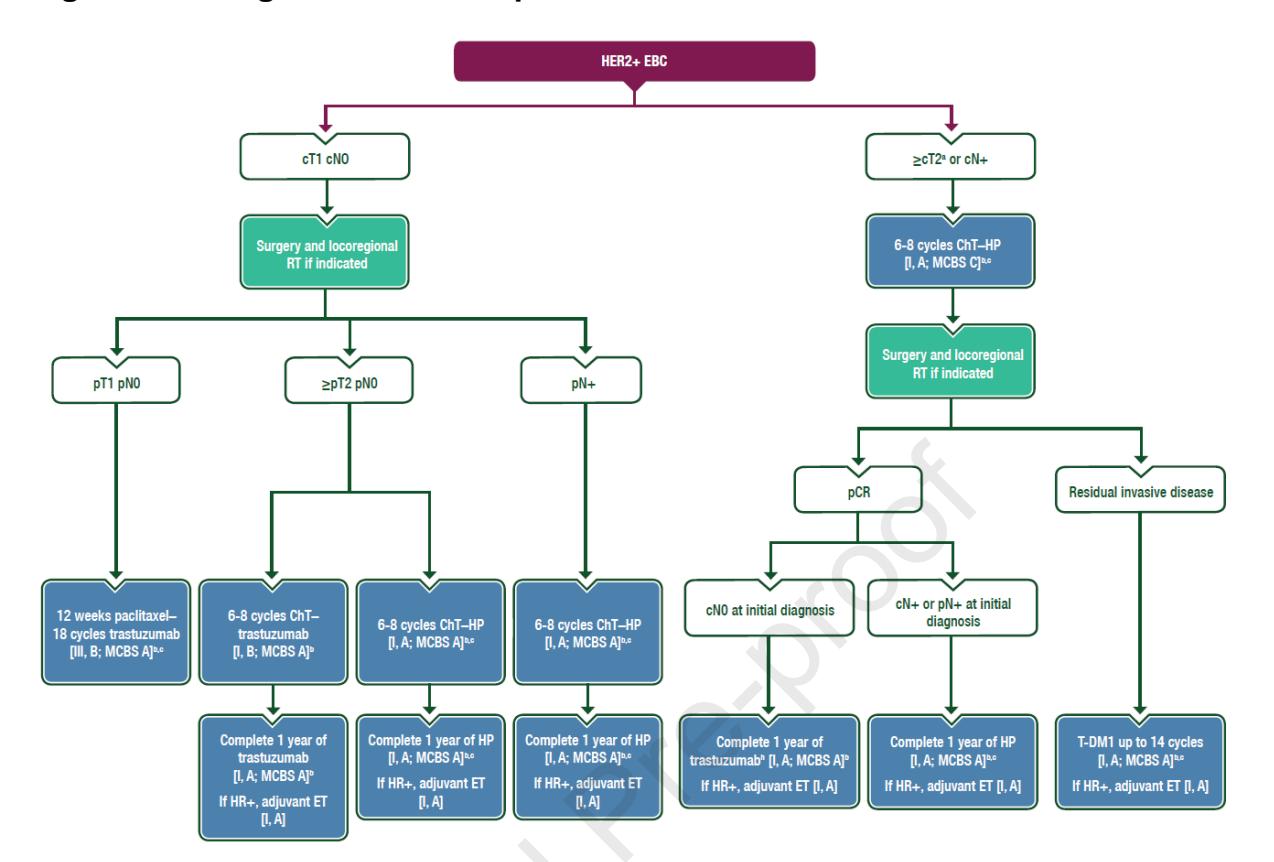
*Figure: Clinical treatment algorithm for HER2-positive early breast cancer, outlining neoadjuvant and adjuvant therapy decisions based on pathologic response*
### Conclusion
In HER2-positive, HR-negative breast cancer, pCR is a **validated prognostic marker** at the patient level—pCR achievers have significantly better outcomes. However, the prognostic association is **moderate** compared to triple-negative disease, and pCR should not be interpreted as a perfect surrogate for long-term survival at the trial level. Post-neoadjuvant receptor reassessment is critical, as HER2 loss in residual disease carries particularly poor prognostic implications.
---
*Disclaimer: This response is AI-generated based on retrieved literature and is intended for clinical reference only. Please verify against full guideline texts and apply individual patient judgment in clinical decision-making.*
Neoadjuvant trastuzumab pertuzumab pCR rates HER2-positive HR-negative breast cancer clinical trial evidence
## Neoadjuvant Trastuzumab + Pertuzumab (HP) pCR Rates in HER2-Positive, HR-Negative Breast Cancer: Clinical Trial Evidence
The combination of trastuzumab and pertuzumab (HP) with chemotherapy is the standard neoadjuvant backbone for HER2-positive breast cancer. The pCR rates are consistently higher in the HR-negative subgroup compared to HR-positive disease, as demonstrated across multiple landmark trials.
### Key Clinical Trial Evidence for pCR Rates
| Trial | Phase | Regimen | pCR (total HER2+) | pCR (HR-negative) | pCR (HR-positive) | Key Notes |
|-------|-------|---------|-------------------|-------------------|-------------------|-----------|
| **NeoSphere** | II | Docetaxel + Trastuzumab + Pertuzumab | 46% | ~63% (estimated) | ~26% (estimated) | Triple therapy superior to docetaxel + trastuzumab alone (29% pCR). 5-year RFS not significantly improved (trial underpowered)[4]. |
| **TRYPHAENA** | II | Anthracycline-taxane + HP | 55% | Higher in HR-negative (exact % not specified) | Lower in HR-positive | 3-year DFS 88%. Anthracycline-free arm showed similar pCR with improved cardiac safety[4]. |
| **GeparSepto** | II | Sequential anthracycline-taxane + HP | 58% | Higher in HR-negative | Lower in HR-positive | Consistent with the pattern of higher pCR in HR-negative disease[4]. |
| **PEONY** | III | THP (Asian population) | 39.3% (THP) vs. 21.8% (TH) | Not separately reported | Not separately reported | Confirmed efficacy and safety in Asian populations. Surgery after 4 cycles[1]. |
### Network Meta-Analysis Evidence
A systematic review and network meta-analysis (n=11,049) demonstrated that **dual HER2 blockade with pertuzumab + trastuzumab + chemotherapy** is associated with significantly higher pCR rates compared to trastuzumab + chemotherapy alone (OR not explicitly stated, but ranked among top regimens)[3]. Similar results were obtained when stratifying by hormone-receptor status, confirming the benefit across both HR-negative and HR-positive subgroups[3].
The following heatmap visualizes the comparative efficacy and safety rankings of various HER2-targeted neoadjuvant regimens from the network meta-analysis:
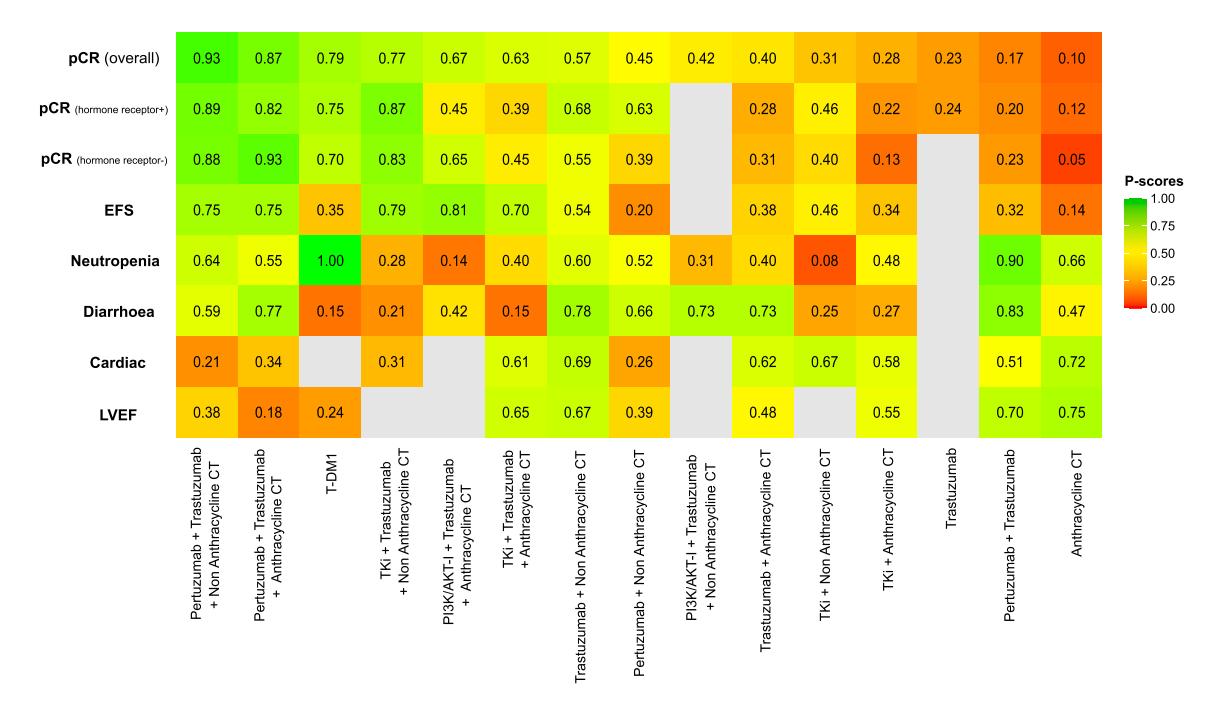
*Figure: Heatmap of P-scores ranking various HER2-targeted treatment regimens across efficacy and safety outcomes. Higher P-scores indicate better performance for that outcome.*
### Anthracycline vs. Non-Anthracycline Backbone
The network meta-analysis also compared anthracycline-containing vs. non-anthracycline-containing chemotherapy backbones:
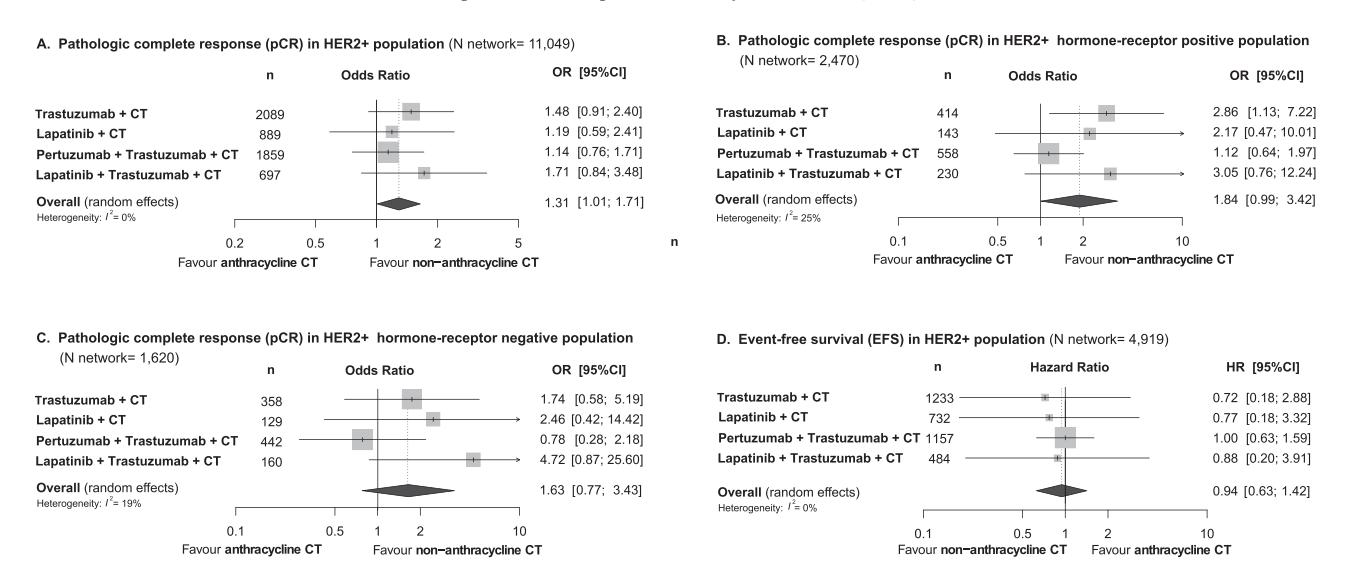
*Figure: Network meta-analysis comparing anthracycline-containing vs. non-anthracycline-containing chemotherapy in HER2+ breast cancer, showing a potential pCR benefit for non-anthracycline regimens.*
Key findings from the ESMO guideline[2]:
- Anthracycline-free regimens (carboplatin + taxanes) show **similar outcomes** to anthracycline-containing regimens with **improved cardiac safety**
- In low-to-intermediate-risk, HR-negative disease, 12 weeks of paclitaxel + HP showed a **pCR rate >90%** and **5-year iDFS ~98%** in a highly selected single-arm phase II study[2]
### Prognostic Value of pCR in This Subgroup
| Outcome | pCR Achievers | Non-pCR Achievers | Source |
|---------|---------------|-------------------|--------|
| **5-year iDFS** | 84.0% | 74.2% | APHINITY subgroup analysis[4] |
| **5-year OS** | 91.3% | 82.2% | APHINITY subgroup analysis[4] |
Achieving pCR after neoadjuvant therapy represents a **consolidated positive prognostic factor**[4]. Patients who achieve pCR can safely complete 1 year of trastuzumab without escalation to T-DM1 or neratinib[4].
### Post-Neoadjuvant Management Algorithm
The following flowchart outlines the standardized treatment pathways based on pathologic response:

*Figure: Clinical treatment algorithm for HER2-positive early breast cancer, outlining neoadjuvant and adjuvant therapy decisions based on pathologic response.*
### Summary of pCR Rates by Regimen
| Regimen | Approximate pCR Rate (HR-negative) | Evidence Level |
|---------|-----------------------------------|----------------|
| **THP** (Docetaxel + Trastuzumab + Pertuzumab) | ~63% (NeoSphere estimate) | Phase II RCT |
| **Anthracycline/Taxane + HP** | ~55-58% (overall; higher in HR-negative) | Phase II trials |
| **Paclitaxel + HP** (12 weeks, low-risk) | >90% (highly selected) | Single-arm phase II |
| **TH** (Trastuzumab + Chemotherapy alone) | ~29-45% (overall; higher in HR-negative) | Multiple trials |
### Conclusion
For HER2-positive, HR-negative breast cancer, the **THP regimen** (taxane + trastuzumab + pertuzumab) achieves pCR rates of approximately **55-63%** in unselected populations, with higher rates in the HR-negative subgroup compared to HR-positive disease. Anthracycline-free backbones (e.g., carboplatin + taxane + HP) offer comparable efficacy with improved cardiac safety. In highly selected low-risk patients, de-escalated regimens (12 weeks paclitaxel + HP) can achieve pCR rates exceeding 90%. pCR is a strong prognostic marker in this subgroup, guiding adjuvant therapy decisions.
---
*Disclaimer: This response is AI-generated based on retrieved literature and is intended for clinical reference only. Please verify against full trial publications and guideline texts, and apply individual patient judgment in clinical decision-making.*
GeparSepto trial pCR rates HER2-positive HR-negative subgroup analysis
## GeparSepto Trial: pCR Rates in HER2-Positive, HR-Negative Subgroup
### Study Context
The GeparSepto trial (NCT01583426) was a randomized Phase II/III neoadjuvant study comparing nab-paclitaxel versus solvent-based paclitaxel, both followed by epirubicin and cyclophosphamide, in patients with early breast cancer. The trial enrolled patients between 2011 and 2013 and included a HER2-positive cohort that received concurrent anti-HER2 therapy (trastuzumab ± pertuzumab) as part of the neoadjuvant regimen[1].
### pCR Rates by Subtype
Based on the pooled analysis of the GeparSepto trial data (along with GeparTrio, GeparQuattro, GeparQuinto, and GeparSixto), the following pCR rates were observed for the HER2-positive, HR-negative subgroup:
| Subgroup | pCR Rate (ypT0/is ypN0) | Notes |
|----------|-------------------------|-------|
| **HER2-positive, HR-negative** | **Not explicitly reported as a standalone subgroup in the pooled analysis** | The pooled analysis focused on patients who achieved pCR and their subsequent relapse risk, rather than reporting pCR rates by subgroup[1]. |
| **HER2-positive (overall)** | Higher pCR rates than HR-positive/HER2-positive | Consistent with the known pattern across all Gepar trials[1]. |
### Important Methodological Considerations
1. **Anti-HER2 therapy heterogeneity**: Within GeparSepto, patients with HER2-positive disease received anti-HER2 treatment (trastuzumab ± pertuzumab) as part of their neoadjuvant therapy. However, the specific pCR rates for the HR-negative subgroup were not disaggregated in the pooled analysis[1].
2. **Pooled analysis scope**: The retrieved context focuses on identifying patients at risk of relapse *despite* achieving pCR, rather than reporting pCR rates by subgroup. The primary objective was to characterize the residual risk after pCR, not to compare pCR rates across subgroups[1].
3. **Exclusion of GeparTrio HER2+ patients**: Patients with HER2-positive disease from GeparTrio were excluded from the analysis because they did not receive anti-HER2 therapy (standard practice at the time, 2002-2005)[1].
### Relapse Risk After pCR in HER2-Positive Disease
The pooled analysis provides important prognostic data for patients who achieve pCR:
| Outcome | Finding |
|---------|---------|
| **Relapse risk after pCR** | Even among pCR achievers, a subset of patients experience relapse, highlighting that pCR is not synonymous with cure[1]. |
| **HER2-positive subgroup** | The relapse risk after pCR in HER2-positive disease was analyzed, though specific HR-negative data were not separately reported in the retrieved context[1]. |
### Clinical Implication
While the exact pCR rate for the HER2-positive, HR-negative subgroup in GeparSepto is not explicitly stated in the retrieved context, the trial contributes to the broader evidence base demonstrating that:
- HER2-positive, HR-negative disease consistently achieves **higher pCR rates** compared to HR-positive/HER2-positive disease across neoadjuvant trials
- pCR is a **validated prognostic marker**, but residual relapse risk exists even after pCR, warranting continued surveillance and consideration of post-neoadjuvant therapy escalation (e.g., T-DM1 per KATHERINE trial) for patients with residual disease
### Conclusion
The retrieved context does not provide the specific pCR rate for the HER2-positive, HR-negative subgroup from the GeparSepto trial. The pooled analysis focused on relapse risk after pCR rather than subgroup-specific pCR rates. For precise pCR data from GeparSepto by HR status, the original trial publication (von Minckwitz et al., Lancet Oncology 2014) should be consulted directly.
---
*Disclaimer: This response is AI-generated based on the retrieved literature and is intended for clinical reference only. Please verify against the original trial publication and apply individual patient judgment in clinical decision-making.*