Postmenopausal Uterine Fibroid Surgery
A 46-year-old female, currently in the postmenopausal stage, has a history of open abdominal surgery for uterine fibroid removal. She continues to have uterine fibroids, a condition she has had for many years. To alleviate menopausal symptoms, is it appropriate for her to undergo hormone therapy?
**Conclusion**: Menopausal hormone therapy (MHT) is generally appropriate for this patient to alleviate vasomotor symptoms, provided a thorough risk-benefit assessment is performed and no absolute contraindications are present. The history of uterine fibroids and prior myomectomy requires specific consideration but is not an absolute contraindication.
---
## Treatment Strategy & Safety Assessment
### Indication for MHT
MHT is the most effective therapy for managing menopause-related vasomotor symptoms (VMS) [1][2][4]. The retrieved guidelines consistently support its use in symptomatic postmenopausal women under 60 years of age or within 10 years of menopause, where the risk-benefit ratio is generally favorable [4][8]. At 46 years old, this patient falls well within this window.
### Uterine Fibroids and MHT: Risk Assessment
The patient's history of uterine fibroids and prior myomectomy is a key consideration. The retrieved evidence does **not** list uterine fibroids as an absolute contraindication to MHT [1][2][3]. However, fibroids are estrogen-sensitive, and MHT could theoretically stimulate growth or reactivate symptoms (e.g., bleeding, bulk symptoms).
**Key Clinical Points:**
- **Not an absolute contraindication**: Current guidelines (NCCN, Korean Society of Menopause, Asia-Pacific Consensus) do not include fibroids in the list of contraindications [1][2][3][4].
- **Monitoring required**: If MHT is initiated, close clinical and ultrasound surveillance of fibroid size and symptoms is warranted.
- **Regimen considerations**: A continuous combined estrogen-progestin regimen (for a patient with an intact uterus) may be preferred over sequential therapy to minimize endometrial stimulation and potential bleeding, which could complicate fibroid assessment.
### Contraindication Screening (Absolute)
Before initiating MHT, the following absolute contraindications must be ruled out [1][2][3]:
- **History of hormonally mediated cancers** (e.g., breast cancer, high-risk endometrial cancer)
- **Unexplained vaginal bleeding** (requires evaluation, e.g., endometrial biopsy)
- **Active or recent thromboembolic event** (VTE, PE, stroke)
- **Active liver disease**
- **Pregnancy**
### Pre-Treatment Evaluation
Based on the retrieved guidelines and consensus statements, the following assessments are recommended before initiating MHT [3][7]:
**The following flowchart outlines the mandatory baseline examinations and follow-up protocols required before and during MHT:**
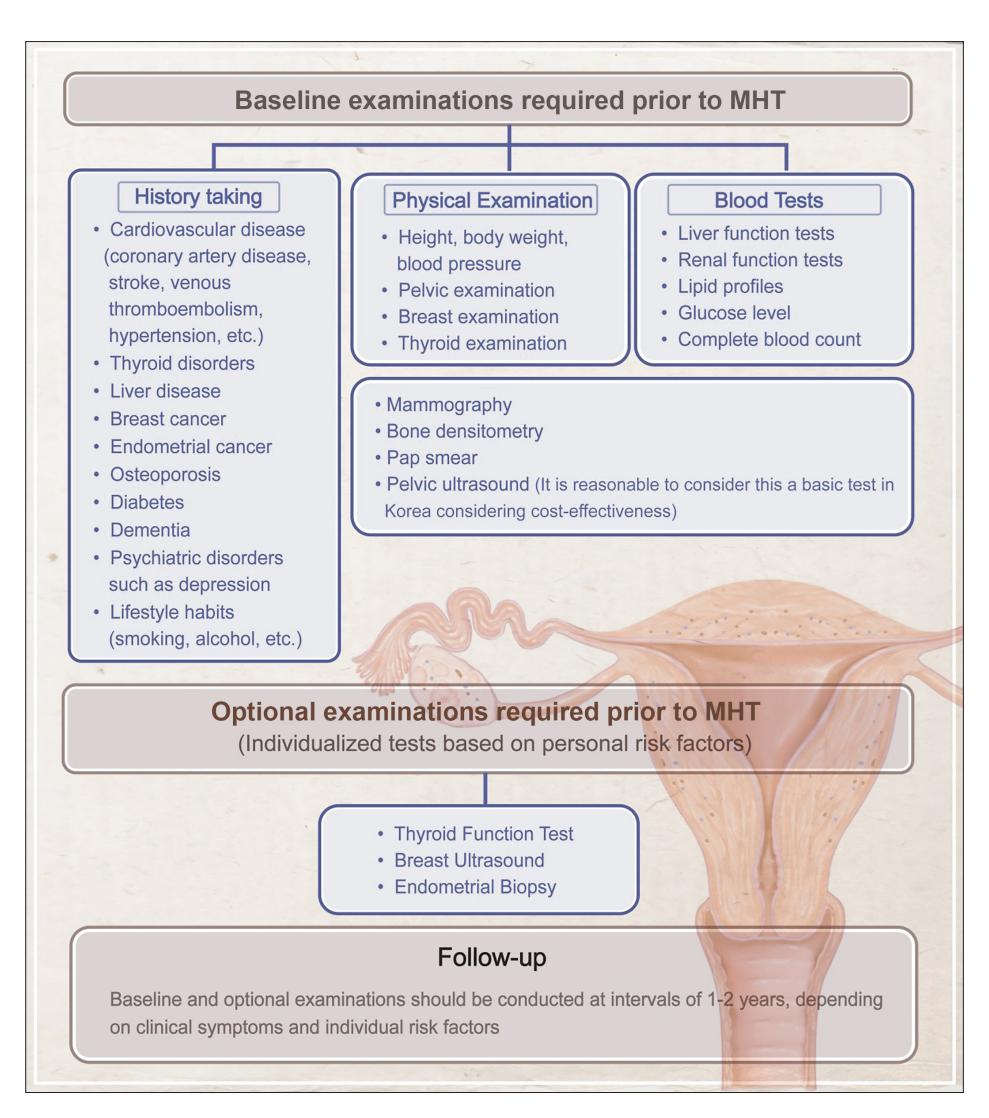
*Figure: This flowchart outlines the mandatory baseline examinations, optional risk-based tests, and follow-up protocols required for patients before and during Menopausal Hormone Therapy (MHT).*
**Mandatory Baseline Assessments:**
1. **Routine blood chemistry** (including lipid profile, liver function, renal function)
2. **Mammography**
3. **Transvaginal ultrasound** (to assess endometrial thickness and fibroid characteristics—size, number, location)
4. **Cardiovascular risk assessment** (blood pressure, BMI, waist-to-hip ratio, smoking status)
**The following comprehensive algorithm illustrates the overall management pathway for perimenopausal and menopausal symptoms:**
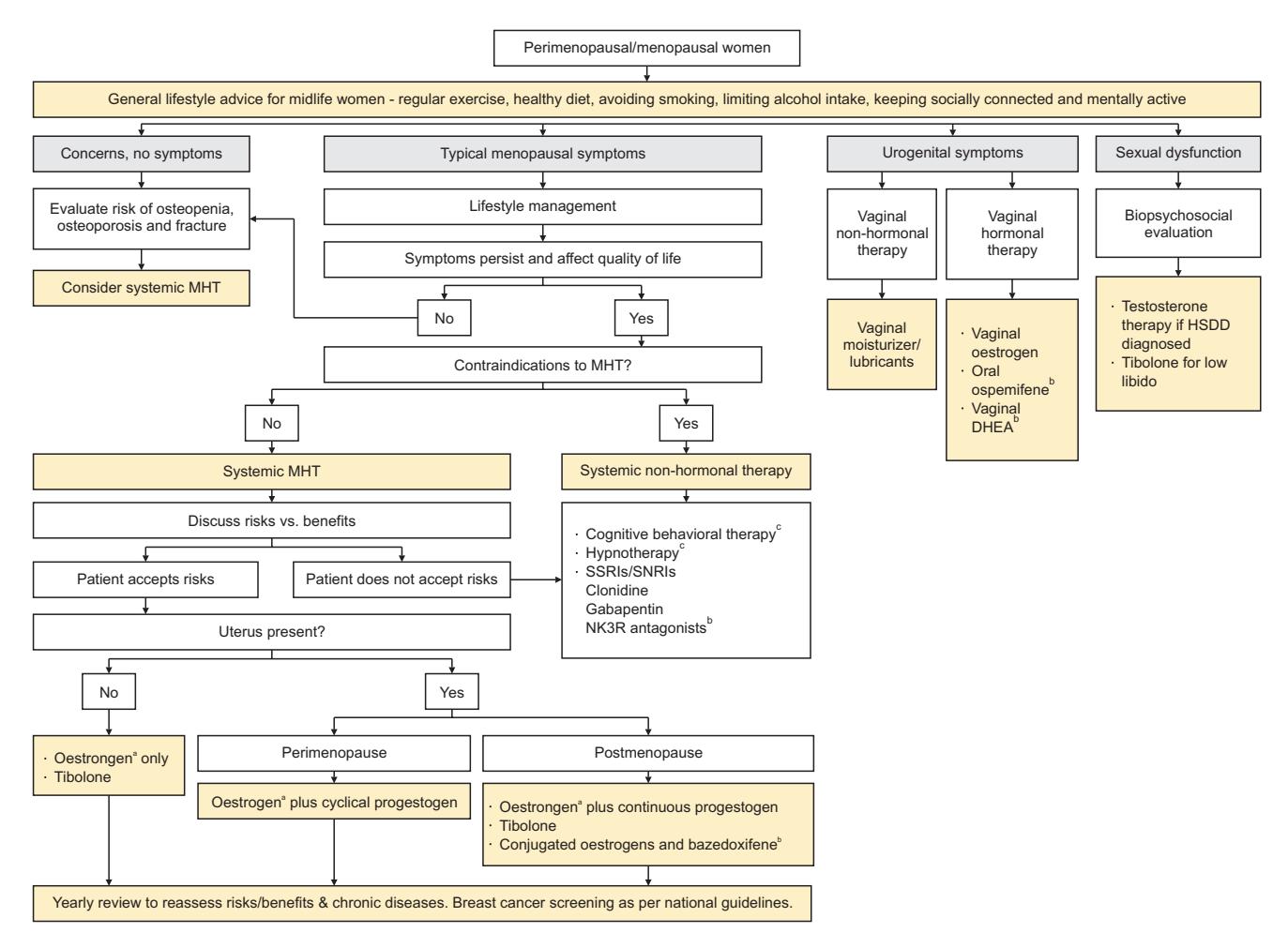
*Figure: A comprehensive clinical flowchart for the management of perimenopausal and menopausal symptoms, outlining lifestyle, hormonal, and non-hormonal treatment pathways.*
### MHT Regimen Selection
- **For a patient with an intact uterus**: A **combined estrogen-progestin** regimen is required to prevent endometrial hyperplasia [1][2].
- **Formulations**: Options include oral, transdermal (patch, gel), and vaginal ring [1][2]. Transdermal estrogen may carry a lower risk of VTE compared to oral formulations.
- **Dosing**: Use the **lowest effective dose** to control symptoms [1][2].
- **Specialist referral**: The NCCN guidelines recommend referral to an appropriate specialist for MHT dosing and management [1][2].
### Special Consideration: Prior Myomectomy
The patient's history of open myomectomy does not contraindicate MHT. However, the presence of residual or recurrent fibroids necessitates:
- **Baseline transvaginal ultrasound** to document fibroid characteristics.
- **Informed consent** discussing the small risk of fibroid growth or symptom recurrence.
- **Regular follow-up** (at least annually) with clinical and ultrasound assessment [4].
### Guideline Pathway Summary
The following summarizes the standard treatment pathway based on retrieved guidelines:
| Step | Action | Evidence Source |
|------|--------|----------------|
| 1 | Confirm indication: Symptomatic VMS in a woman <60 yrs / <10 yrs post-menopause | [4][8] |
| 2 | Screen for absolute contraindications (breast cancer, VTE, liver disease, etc.) | [1][2][3] |
| 3 | Perform baseline assessments (mammography, TVUS, blood work, CV risk) | [3][7] |
| 4 | Initiate MHT at lowest effective dose; combined regimen for intact uterus | [1][2] |
| 5 | Monitor annually: clinical exam, imaging, risk reassessment | [4] |
---
### Further Diagnostic Suggestions (Reference Only)
To enable a more accurate assessment, if available, please consider providing:
- **Transvaginal ultrasound results**: To assess current fibroid size, number, and endometrial thickness, which directly impacts the risk-benefit calculus.
- **Personal and family history of breast cancer or VTE**: To definitively rule out absolute contraindications.
---
*Disclaimer: This analysis is based on retrieved clinical guidelines and consensus statements. Clinical decision-making should be individualized, and all recommendations should be verified against the latest official drug labeling and local regulatory approvals. The final treatment plan requires shared decision-making between the clinician and the patient.*