Lower Extremity Venous Thrombosis
Is it appropriate to treat left lower extremity venous thrombosis (involving multiple thrombi in the popliteal and posterior tibial veins, with the largest measuring 62 mm × 6 mm) by infusing 400,000 units of urokinase via a superficial vein in the left foot (administered for 2 hours, twice daily), in combination with anticoagulation using rivaroxaban 15 mg twice daily?
Based on the retrieved evidence, the proposed regimen of **superficial foot vein infusion of urokinase 400,000 IU twice daily combined with rivaroxaban 15 mg twice daily** for acute left lower extremity venous thrombosis is **not appropriate** and deviates from standard guideline-directed therapy in several critical aspects.
---
## Drug Safety & Treatment Strategy Analysis
### 1. Anticoagulation Regimen: Rivaroxaban Dosing Error
The proposed rivaroxaban dose of **15 mg twice daily** is incorrect for the treatment phase of acute DVT/PE.
- **Correct Acute Phase Dosing**: According to the rivaroxaban drug label [2] and the NCCN Guidelines (V.3.2025) [3], the approved regimen for the treatment of DVT and/or PE is:
- **Days 1–21**: **15 mg orally twice daily** with food.
- **Day 22 onwards**: **20 mg orally once daily** with food for the remaining treatment duration.
- **Error Identification**: The user's regimen uses the 21-day loading dose (15 mg BID) as a maintenance dose without specifying a transition to the standard 20 mg once-daily maintenance dose. Prolonged use of the 15 mg BID regimen beyond 21 days is not supported by the drug label and may represent an unnecessary dosing complexity without proven benefit over the standard once-daily maintenance dose.
### 2. Thrombolytic Strategy: Urokinase via Superficial Foot Vein
The proposed method of administering urokinase (400,000 IU over 2 hours, twice daily) via a superficial vein in the left foot is **not a standard or guideline-recommended approach** for acute proximal DVT.
- **Guideline-Recommended Approach**: For acute (≤14 days) symptomatic, extensive proximal DVT (such as iliofemoral DVT), the 2026 AHA/ACC Guideline [1] and the AHA Scientific Statement [6] suggest that **catheter-directed thrombolysis (CDT)** or **pharmacomechanical catheter-directed thrombolysis (PCDT)** may be considered in select patients with good functional status, low bleeding risk, and ≥1-year life expectancy [Class IIb, Level B] [6].
- **Route of Administration**: CDT/PCDT involves delivering the thrombolytic agent directly into the thrombus via a multi-sidehole catheter placed under imaging guidance. This maximizes local drug concentration and minimizes systemic exposure.
- **Why Superficial Foot Vein Infusion is Inappropriate**:
- **Ineffective Delivery**: Infusing urokinase into a superficial foot vein will result in the drug entering the deep venous system via perforators, but it will be significantly diluted and will not achieve therapeutic concentrations within the occlusive thrombus in the popliteal and posterior tibial veins. The drug will largely be shunted through patent veins.
- **Increased Systemic Risk**: This method effectively constitutes systemic thrombolysis, which carries a high risk of major bleeding (including intracranial hemorrhage) without the targeted efficacy of CDT.
- **Guideline Prohibition**: Systemic thrombolysis is **not recommended** for the treatment of DVT [Class III, Level A] [6]. The proposed superficial foot vein infusion is functionally a form of systemic thrombolysis and is therefore contraindicated by this high-level evidence.
### 3. Urokinase Dosing and Regimen
- **Standard Dosing**: The AWMF S2k Guideline [7] lists standard systemic thrombolysis regimens for acute PE, including urokinase: **4,400 IU/kg over 10 minutes, followed by 4,400 IU/kg/h for 12–24 hours** or an accelerated regimen of **3 million IU over 2 hours** [7].
- **Comparison**: The proposed dose of 400,000 IU over 2 hours is significantly lower than standard systemic regimens. For a 70 kg patient, the standard loading dose alone would be 308,000 IU, followed by a continuous infusion. The proposed regimen is neither a standard systemic dose nor a catheter-directed dose. It represents an unvalidated, sub-therapeutic approach that combines the risks of systemic thrombolysis with questionable efficacy.
### 4. Patient Selection and Contraindication Screening
The query does not provide critical patient information required before initiating any thrombolytic therapy.
- **Absolute Contraindications to Thrombolysis**: Active bleeding, recent major surgery/trauma (within 3 weeks), history of intracranial hemorrhage, recent ischemic stroke (within 3 months), known bleeding diathesis, severe uncontrolled hypertension.
- **Thrombus Extent and Timing**: The thrombus involves the popliteal and posterior tibial veins. This is a **distal** (below-knee) DVT, not an extensive **proximal** (iliofemoral) DVT. The 2026 AHA/ACC Guideline [1] and NCCN Guidelines [3] recommend anticoagulation for distal DVT unless a contraindication exists. The role of thrombolysis for isolated distal DVT is even more limited and not routinely recommended.
- **Cancer Status**: The NCCN Guidelines [3] provide specific recommendations for cancer-associated thrombosis, where DOACs (including rivaroxaban) are preferred for patients without gastric/gastroesophageal lesions. The patient's cancer status is unknown.
### 5. Special Population Considerations
- **Renal Function**: The retrieved evidence [4] specifically raises concerns about rivaroxaban use in patients with advanced kidney disease (CrCl 15-29 mL/min). The high loading doses (15 mg BID) are a particular concern, and expert consensus did not include rivaroxaban as an option for VTE treatment in this population [4]. The patient's renal function is not provided.
---
## Guideline Recommendations Summary
| Recommendation | Source | Class/Level |
|---|---|---|
| Anticoagulation is recommended for distal DVT (popliteal/posterior tibial) unless contraindicated. | NCCN V.3.2025 [3] | Standard of Care |
| For acute DVT treatment, use rivaroxaban 15 mg BID for 21 days, then 20 mg daily. | Drug Label [2], NCCN [3] | Definitive |
| Systemic thrombolysis is **not recommended** for DVT. | AHA Statement [6] | **Class III, Level A** |
| CDT/PCDT may be considered for select patients with acute (≤14 days) symptomatic, extensive **proximal** DVT. | AHA Statement [6] | Class IIb, Level B |
| Reduced-dose DOACs (apixaban 2.5 mg BID or rivaroxaban 10 mg daily) may be considered for extended therapy (>3-6 months) in patients not at high risk of recurrence. | ESVS 2023 [8] | Class IIa, Level B |
---
**The following flowchart illustrates the evidence-based antithrombotic management of proximal DVT:**
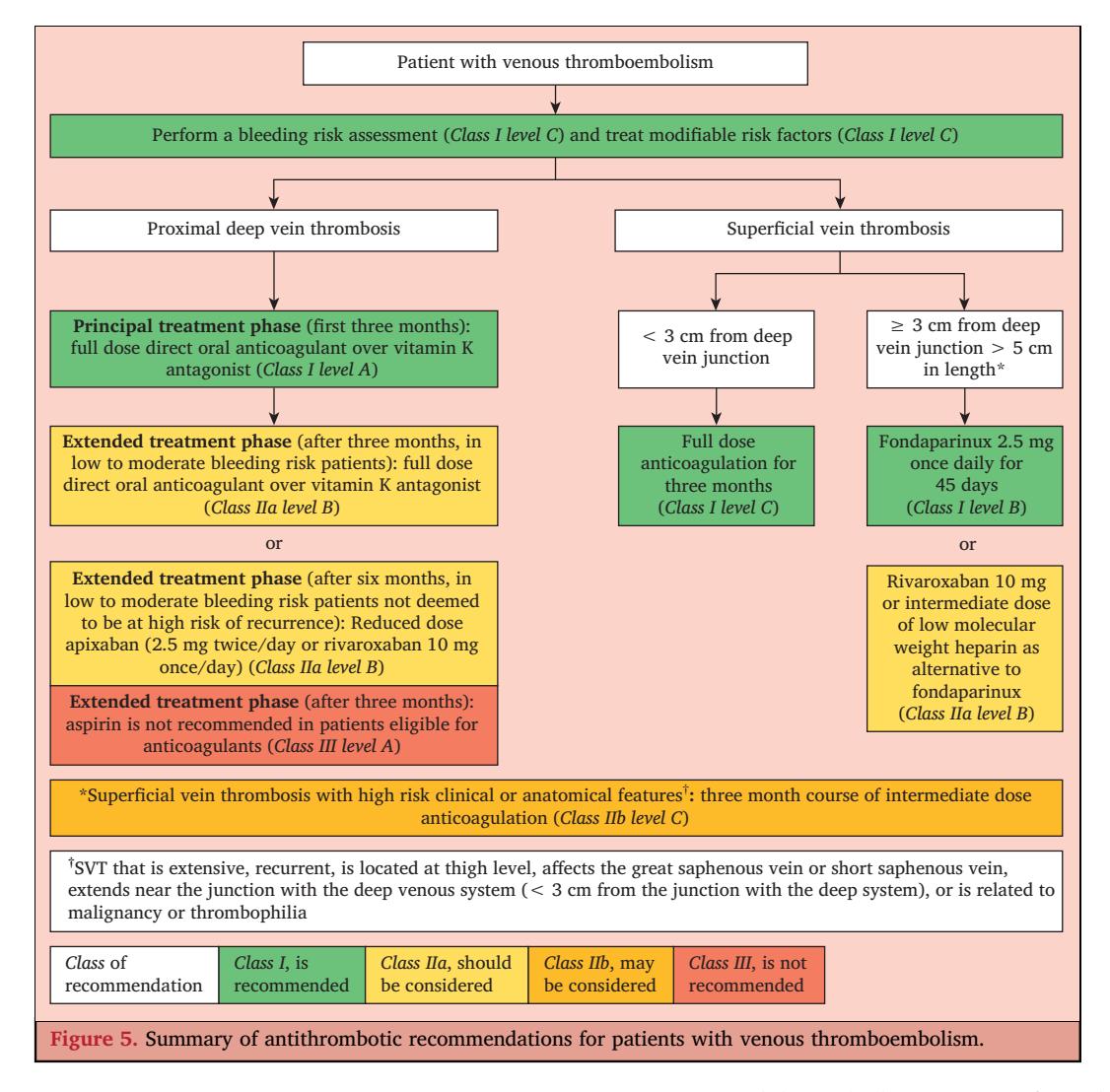
*Figure: Evidence-based clinical flowchart for the antithrombotic management of proximal deep vein thrombosis and superficial vein thrombosis, detailing drug selection, dosing, and duration.*
**The following NCCN algorithm outlines the management of acute DVT based on anatomical location:**

*Figure: This NCCN guideline flowchart outlines the management of acute deep vein thrombosis (DVT) based on anatomical location and the presence of contraindications to anticoagulation.*
---
## Conclusion
The proposed regimen is **not appropriate**. The correct standard of care for this patient (acute distal DVT of the popliteal and posterior tibial veins) is **therapeutic anticoagulation alone** with a DOAC (e.g., rivaroxaban 15 mg BID for 21 days, then 20 mg daily). The use of urokinase via a superficial foot vein is an unvalidated, potentially harmful approach that constitutes systemic thrombolysis, which is explicitly **not recommended** for DVT [Class III, Level A] [6]. Thrombolysis (CDT/PCDT) should only be considered in highly selected patients with acute, symptomatic, extensive **proximal** (iliofemoral) DVT, and must be delivered via a catheter directly into the thrombus.
---
### Further Diagnostic Suggestions (Reference Only)
To enable a more accurate assessment, if available, please consider providing:
* **Renal function (CrCl/eGFR)**: Essential for safe rivaroxaban dosing and to assess bleeding risk.
* **Bleeding risk assessment**: History of active bleeding, recent surgery/trauma, or bleeding diathesis.
* **Symptom duration**: To confirm the thrombus is acute (≤14 days) if thrombolysis is being considered.
* **Cancer status**: To guide DOAC vs. LMWH selection per NCCN guidelines.
---
*This analysis is based on retrieved evidence from 2023-2026 guidelines and drug labels. Clinical decisions should be individualized based on patient-specific factors and verified against the latest official prescribing information.*