Dapagliflozin in HFrEF Low eGFR
For HFrEF patients starting the “foundational four”, is dapagliflozin still recommended when eGFR is below 30 ml/min/1.73m²?
**Yes, dapagliflozin is recommended for HFrEF patients with an eGFR below 30 mL/min/1.73 m², provided it is not initiated below 25 mL/min/1.73 m², and continuation is supported even below 20 mL/min/1.73 m² based on recent label updates and clinical trial data.**
---
## Clinical Evidence & Efficacy
The cardiovascular benefits of SGLT2 inhibitors (SGLT2i) in heart failure with reduced ejection fraction (HFrEF) are preserved across a wide range of renal function, including advanced chronic kidney disease (CKD).
- **Pivotal trial inclusion criteria**: The landmark HFrEF trials for dapagliflozin (DAPA-HF) and empagliflozin (EMPEROR-Reduced) enrolled patients with eGFR as low as 20–30 mL/min/1.73 m². Importantly, participants whose eGFR fell below 20 mL/min/1.73 m² during the trials **continued treatment** and derived considerable benefit [3].
- **Renal and cardiovascular protection**: The committee for the NICE guideline [NG28] noted that the cardiovascular and renal protective effects of SGLT2i are retained at eGFR values below 30 mL/min/1.73 m², even though the glycemic benefits may diminish or be absent [1].
- **Expected eGFR dip**: A transient drop in eGFR is expected upon SGLT2i initiation due to increased afferent glomerular arteriolar tone. Clinicians should **not** adjust or discontinue the medication solely based on this expected change [3].
## Dosing & Administration
The initiation threshold for dapagliflozin has been updated in recent regulatory labels and guidelines.
| Source | Initiation Threshold | Continuation Below Threshold |
|--------|---------------------|------------------------------|
| **2024 ACC Expert Consensus** | Lower limit of **25 mL/min/1.73 m²** for dapagliflozin [3] | Supported; trial participants continued below 20 mL/min/1.73 m² with benefit [3] |
| **2022 CCS Guidelines** | Initiation not recommended for eGFR < **25 mL/min/1.73 m²** [5] | N/A |
| **UK Kidney Association** | Not recommended to initiate if eGFR < **15 mL/min/1.73 m²** [6] | N/A |
| **ADA/KDIGO Consensus** | Approved for use at 10 mg once daily with eGFR **25 to <45 mL/min/1.73 m²** [4] | Higher dose not effective for glucose lowering but CV benefit preserved [4] |
**Standard dosing for HFrEF**: Dapagliflozin **10 mg orally once daily**.
## Safety Profile
### Contraindications and Precautions
- **Absolute contraindications**: End-stage kidney disease (KF) and patients on dialysis [2].
- **Initiation caution**: Do not initiate dapagliflozin when eGFR is persistently <25 mL/min/1.73 m² [3][5].
- **Monitoring**: Closer monitoring of renal function and electrolytes is advised in patients with eGFR <30 mL/min/1.73 m² [3].
### Adverse Effects
- **Common**: Genital mycotic infections, volume depletion (especially with concurrent diuretic use), and the expected initial eGFR dip.
- **Serious but rare**: Euglycemic diabetic ketoacidosis (eDKA), Fournier gangrene (necrotizing fasciitis of the perineum).
## Guideline Recommendations
The following clinical workflow illustrates the initiation and management of SGLT2i in the context of HFrEF and CKD:
**The following flowchart outlines the clinical management pathway for SGLT2 inhibitor use:**
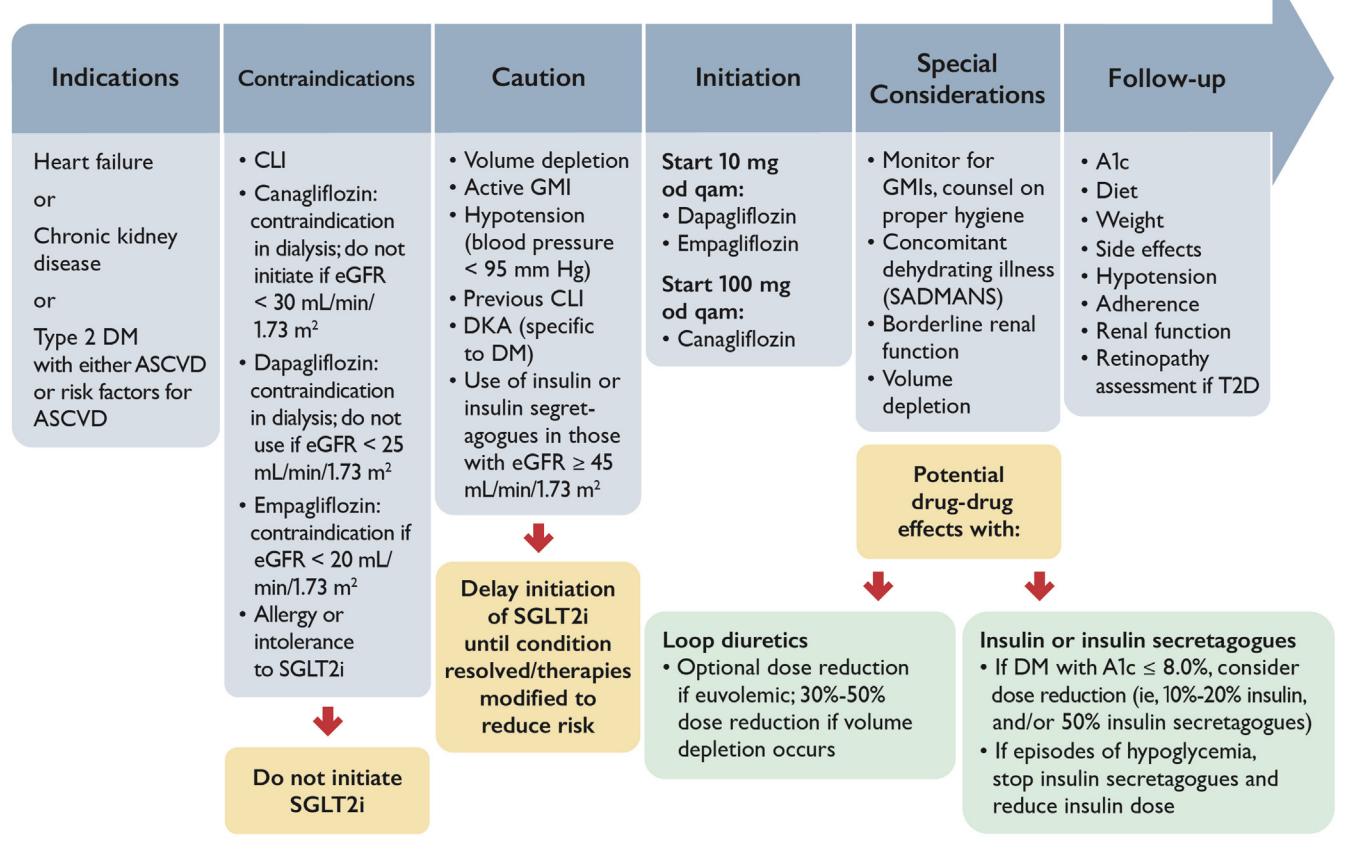
*Figure: A clinical workflow for the initiation and management of SGLT2 inhibitors (SGLT2i), detailing indications, renal-specific contraindications, and dose adjustments for concomitant therapies.*
The quadruple therapy foundation for HFrEF (ARNI/ACEi/ARB, beta-blocker, MRA, SGLT2i) is recommended regardless of baseline eGFR, with the renal-specific caveats noted above.
**The following diagram illustrates the GDMT pathway for Stage C HFrEF:**
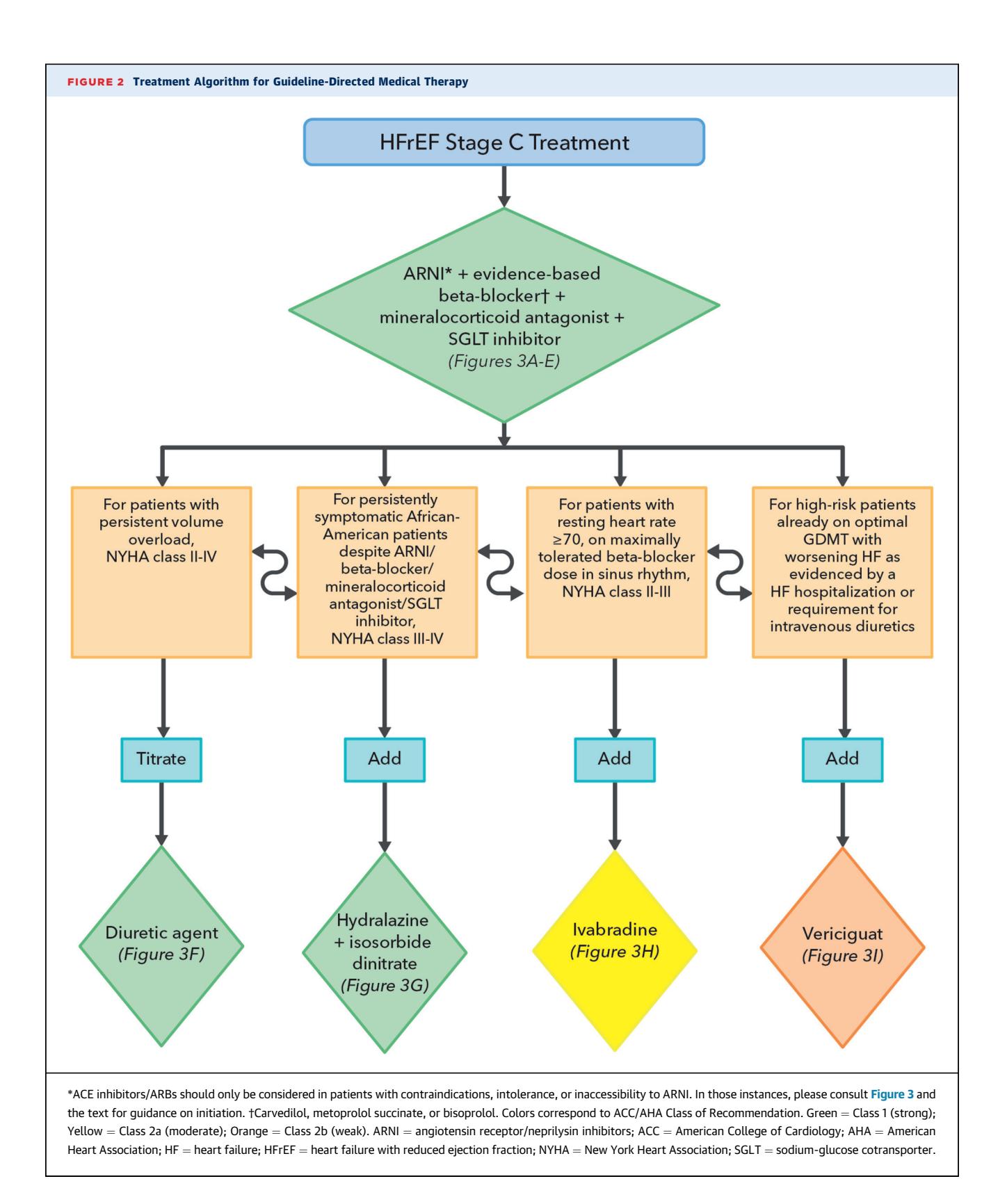
*Figure: This flowchart outlines the guideline-directed medical therapy (GDMT) for Stage C Heart Failure with Reduced Ejection Fraction (HFrEF), prioritizing a quadruple therapy foundation followed by targeted add-on treatments.*
## Critical Gaps
- The available evidence does not provide specific guidance on the optimal monitoring frequency (e.g., serum creatinine, potassium) for patients with eGFR <30 mL/min/1.73 m² on dapagliflozin.
- Long-term safety data (beyond 2–3 years) for SGLT2i in patients with eGFR persistently <25 mL/min/1.73 m² are limited.
## Conclusion
Dapagliflozin is a recommended component of the "foundational four" for HFrEF even when eGFR is below 30 mL/min/1.73 m², with initiation supported down to 25 mL/min/1.73 m² and continuation of therapy below that threshold supported by clinical trial data and recent label updates.
---
*This response is based on retrieved clinical guidelines and expert consensus documents. Always verify against the latest official prescribing information and apply individual patient-specific clinical judgment.*