Lumakras Dosing Guidelines
What is the recommended dosing for Lumakras in KRAS-mutant non-small cell lung cancer?
The recommended dosage of Lumakras (sotorasib) for KRAS G12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) is **960 mg orally once daily** until disease progression or unacceptable toxicity [4].
**The following flowchart illustrates the clinical treatment algorithm for advanced NSCLC, including the positioning of KRAS G12C targeted therapy:**
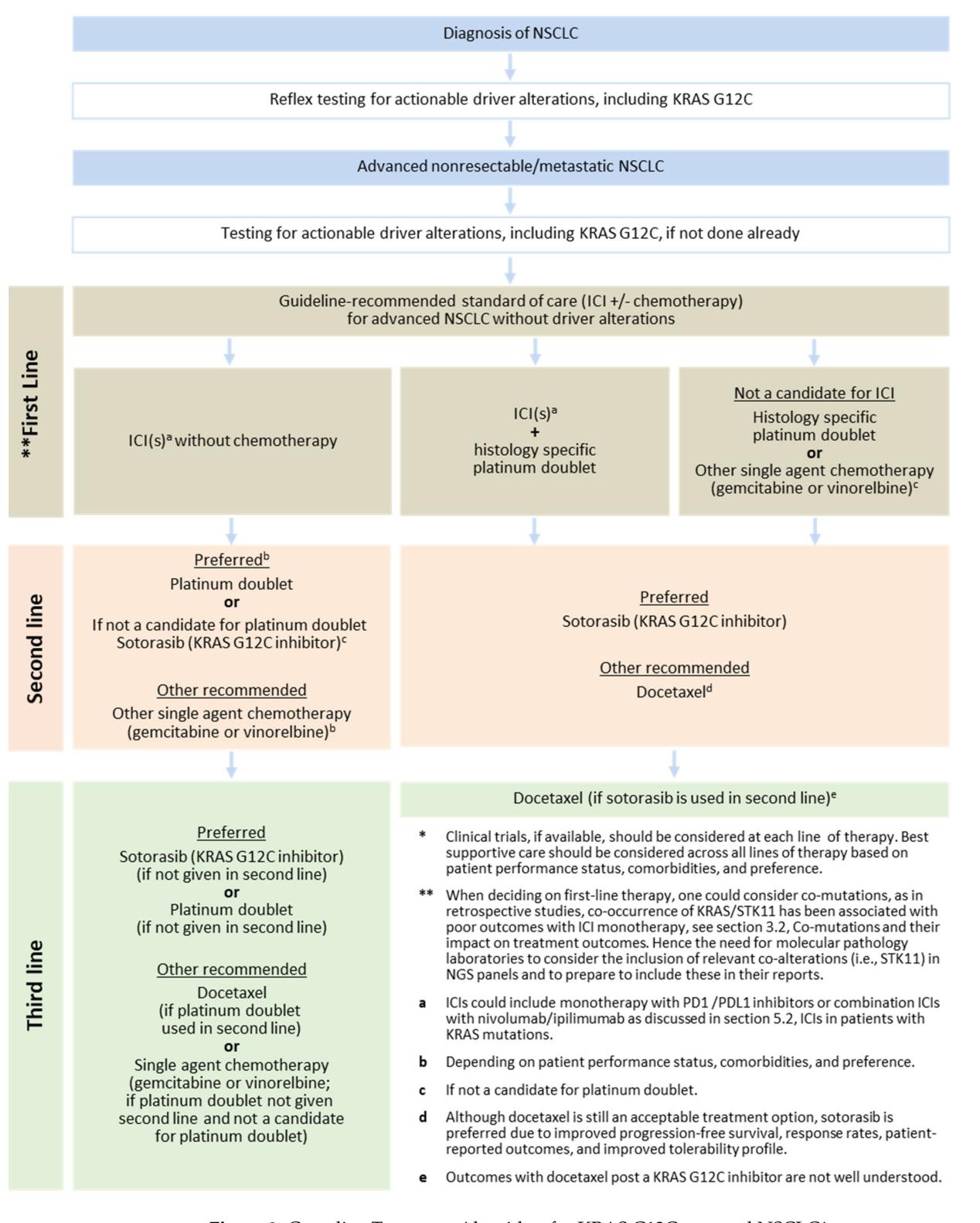
*Figure: A clinical flowchart outlining the treatment pathway for advanced or metastatic NSCLC, detailing the integration of KRAS G12C targeted therapy across first, second, and third-line settings*
## Dosing & Administration Details
- **Formulation**: 120 mg tablets (eight tablets per dose) [4]
- **Route**: Oral [4]
- **Timing**: Take at the same time each day [4]
- **Food Effect**: Can be taken with or without food [4]
- **Tablet Handling**: Swallow tablets whole. Do **not** chew, crush, or split tablets [4]
## Missed Dose & Vomiting Instructions
- **Missed dose**: If a dose is missed by more than 6 hours, skip that dose and take the next dose as prescribed the next day. Do **not** take two doses at the same time to make up for the missed dose [4].
- **Vomiting**: If vomiting occurs after taking Lumakras, do **not** take an additional dose. Resume with the next scheduled dose the following day [4].
## Dose Modifications for Toxicity
Based on the prescribing information, dose modifications are required for specific adverse events [4]:
| Toxicity | Management |
|----------|------------|
| **Hepatotoxicity** | Monitor liver function tests every 3 weeks for the first 3 months, then monthly. Withhold, reduce dose, or permanently discontinue based on severity [4]. |
| **Interstitial Lung Disease (ILD)/Pneumonitis** | Immediately withhold for suspected ILD/pneumonitis. Permanently discontinue if no other cause is identified [4]. |
## Indication Context
This dosing applies to the FDA-approved indication for KRAS G12C-mutated locally advanced or metastatic NSCLC in adults who have received at least one prior systemic therapy. This indication was approved under **accelerated approval** based on overall response rate (ORR) and duration of response (DOR); continued approval may be contingent on confirmatory trial results [1][2][4].
**Important**: A companion diagnostic test is required to confirm the presence of the KRAS G12C mutation before initiating therapy [1][2].
---
*This information is based on retrieved prescribing information and consensus recommendations. Clinicians should verify against the most current full prescribing information and apply individual patient considerations when making treatment decisions.*