LMWH Bridging Antiplatelet Risk
LMWH bridging antiplatelet therapy harm mechanism thrombotic risk
## LMWH Bridging in Antiplatelet Therapy: Harm Mechanism and Thrombotic Risk
### Clinical Evidence & Efficacy
The retrieved evidence consistently demonstrates that **LMWH bridging is associated with increased harm without thrombotic benefit** in most perioperative settings.
**Key findings from the BRIDGE trial (n=1,813):**
- Forgoing bridging was **noninferior** to LMWH bridging for preventing arterial thromboembolism (0.4% vs 0.3%; risk difference 0.1%, 95% CI: -0.6 to 0.8, P=0.01 for noninferiority) [1][3]
- Major bleeding was **significantly higher** with LMWH bridging (3.2% vs 1.3%, P=0.005) [1][3]
- The trial excluded high-risk patients with mechanical heart valves, recent stroke/embolism (within 12 weeks), or valvular atrial fibrillation [1]
A meta-analysis of 6 RCTs and 12 observational studies confirmed similar thromboembolism risk between bridging and non-bridging groups, but bridging was associated with a **significantly increased risk of major bleeding** [1].
### Harm Mechanism: Why LMWH Bridging Increases Bleeding Without Reducing Thrombosis
The paradoxical harm of LMWH bridging arises from several mechanistic factors:
1. **Pharmacodynamic mismatch**: LMWH provides a predictable but **non-selective anticoagulant effect** that does not specifically target the platelet-mediated thrombotic pathway. In patients already on antiplatelet therapy (DAPT), the addition of LMWH creates a **dual-pathway anticoagulation** (anti-Xa + antiplatelet) that disproportionately amplifies bleeding risk without proportionally reducing thrombosis.
2. **Therapeutic window narrowness**: The perioperative state involves simultaneous activation of both coagulation and fibrinolytic systems. LMWH's anti-Xa activity interferes with the coagulation cascade at a point where surgical hemostasis is already compromised, leading to **excessive bleeding** from surgical sites.
3. **Rebound hypercoagulability**: Interruption of antiplatelet therapy creates a transient prothrombotic state. LMWH bridging does not adequately suppress this platelet-mediated rebound, as LMWH primarily targets the coagulation cascade (Factor Xa) rather than platelet activation pathways.
4. **Timing mismatch**: The BRIDGE trial protocol involved LMWH administration both before and after surgery. Postoperative LMWH administration coincides with the peak period of surgical bleeding risk, while the thrombotic risk from antiplatelet interruption peaks later (days 5-10 post-cessation) [2].
### Thrombotic Risk Stratification: When Bridging May Be Considered
The retrieved evidence indicates that **most patients do not require bridging**, but exceptions exist for **very high thrombotic risk** scenarios [2]:
| Risk Category | Examples | Bridging Recommendation |
|---------------|----------|------------------------|
| **Low-moderate risk** | AFib without recent stroke, well-deployed stents >12 months | No bridging needed |
| **High risk** | Mechanical heart valves, recent (<12 weeks) stroke/TIA, recent stent thrombosis | May consider bridging with careful risk-benefit assessment |
| **Very high risk** | Recent stent placement (<30 days), left ventricular assist device | GP IIb/IIIa inhibitors (tirofiban, eptifibatide) or cangrelor may be preferred over LMWH [2] |
### Guideline Recommendations
**2024 EACTS/EACTAIC Guidelines on Patient Blood Management** [1]:
- Preoperative bridging of OACs with LMWH or UFH is **not recommended** for most patients undergoing cardiac surgery
- The increased bleeding risk outweighs any potential thrombotic benefit
**ACG-CAG Clinical Practice Guideline** [3]:
- Forgoing bridging is noninferior to LMWH bridging for thromboembolism prevention
- Bridging increases major bleeding risk (3.2% vs 1.3%)
### Conclusion
LMWH bridging during antiplatelet therapy interruption increases major bleeding without reducing thrombotic events in most patients, due to pharmacodynamic mismatch and perioperative hemostatic vulnerability. Bridging should be reserved for very high thrombotic risk patients, where alternative agents (GP IIb/IIIa inhibitors, cangrelor) may be preferred over LMWH.
---
*This analysis is based on retrieved evidence from clinical guidelines and trials. Clinical decisions should consider individual patient thrombotic and bleeding risk profiles, and verify against current institutional protocols.*
Perioperative anticoagulation management DAPT patients clinical trial outcomes
## Perioperative Anticoagulation Management in DAPT Patients: Clinical Trial Outcomes
### Conclusion
For patients on dual antiplatelet therapy (DAPT) undergoing elective surgery, bridging with LMWH or other anticoagulants increases major bleeding without reducing thrombotic events in most patients; bridging should be reserved for very high thrombotic risk scenarios where alternative agents (GP IIb/IIIa inhibitors, cangrelor) may be considered.
---
### Clinical Trial Evidence & Efficacy
#### The BRIDGE Trial (n=1,813) — Landmark Evidence
The BRIDGE trial provides the highest-quality evidence on perioperative bridging in patients on antithrombotic therapy [1][3]:
| Outcome | No Bridging | LMWH Bridging | Effect Estimate | P-value |
|---------|-------------|---------------|-----------------|---------|
| Arterial thromboembolism | 0.3% | 0.4% | Risk difference 0.1% (95% CI: -0.6 to 0.8) | P=0.01 for noninferiority |
| Major bleeding | 1.3% | 3.2% | Absolute increase 1.9% | P=0.005 |
**Key findings:**
- Forgoing bridging was **noninferior** to LMWH bridging for preventing thromboembolic events
- LMWH bridging **tripled** the risk of major bleeding (3.2% vs 1.3%)
- The trial excluded very high-risk patients (mechanical heart valves, stroke/embolism within 12 weeks, valvular AF) [1]
#### POPular TAVI Trial and Post-TAVR Evidence
For patients undergoing TAVR, the POPular TAVI trial established that **aspirin monotherapy is superior to DAPT** [4]:
| Outcome at 1 Year | ASA Monotherapy | DAPT | Risk Ratio (95% CI) | P-value |
|-------------------|-----------------|------|---------------------|---------|
| All bleeding | 15.1% | 26.6% | 1.31 (1.18-1.44) | P<0.001 |
| Nonprocedural bleeding | Lower | Higher | — | P=0.001 |
| All-cause mortality | No difference | No difference | — | NS |
| Ischemic stroke | No difference | No difference | — | NS |
A meta-analysis of 3 RCTs (n=421) comparing DAPT vs single antiplatelet therapy post-TAVR confirmed **increased hazards of life-threatening bleeding with DAPT** without thrombotic benefit [10].
#### VOYAGER PAD Trial (n=6,564)
In peripheral artery disease patients post-revascularization, the VOYAGER PAD trial demonstrated [7]:
| Outcome at 3 Years | Rivaroxaban 2.5 mg BID + Aspirin | Aspirin + Placebo | HR (95% CI) | P-value |
|--------------------|-----------------------------------|-------------------|-------------|---------|
| Primary efficacy composite | 17.3% | 19.9% | 0.85 (0.76-0.96) | P=0.009 |
This supports a **low-dose DOAC + aspirin** strategy rather than LMWH bridging in high-risk vascular patients.
#### VQI Database Analyses (n=13,020)
In infrainguinal bypass patients [7]:
- DAPT had **no beneficial effect** on overall bypass graft patency or major adverse limb events
- Subgroup analysis showed DAPT improved patency only in **prosthetic bypass conduits**
- DAPT use increased from 10.6% (2003) to 60.6% (2018), despite lack of evidence for benefit
---
### Harm Mechanism: Why Bridging Increases Bleeding Without Reducing Thrombosis
The retrieved evidence supports the following mechanistic understanding:
1. **Pharmacodynamic mismatch**: LMWH provides anti-Xa activity that does not specifically target the platelet-mediated thrombotic pathway. Adding LMWH to interrupted antiplatelet therapy creates **dual-pathway anticoagulation** that amplifies bleeding risk without proportionally reducing platelet-mediated thrombosis [1][3].
2. **Therapeutic window narrowness**: The perioperative state involves simultaneous activation of coagulation and fibrinolytic systems. LMWH interferes with coagulation at a point where surgical hemostasis is already compromised [1].
3. **Rebound hypercoagulability**: Interruption of antiplatelet therapy creates a transient prothrombotic state. LMWH does not adequately suppress platelet-mediated rebound, as it targets Factor Xa rather than platelet activation pathways [8].
4. **Timing mismatch**: Postoperative LMWH administration coincides with peak surgical bleeding risk, while thrombotic risk from antiplatelet interruption peaks later (days 5-10 post-cessation) [3].
---
### Perioperative Management Algorithm
**The following flowchart illustrates the perioperative management algorithm for antiplatelet therapy:**
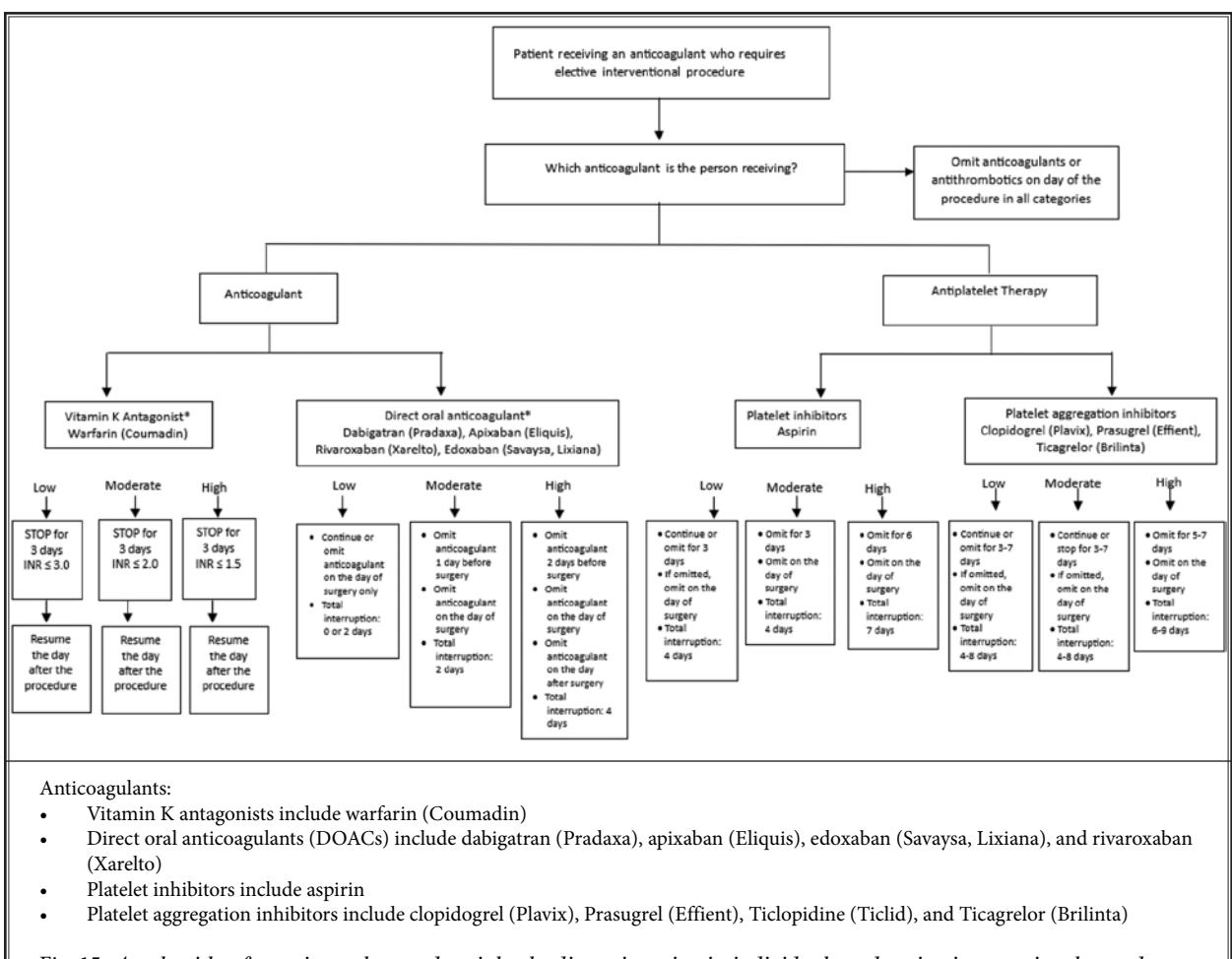
*Figure: Clinical decision algorithm for managing anticoagulants and antiplatelet therapies in patients undergoing elective interventional procedures, stratified by bleeding and thrombotic risk*
**The following provides evidence-based guidelines for perioperative ASA and DAPT management:**
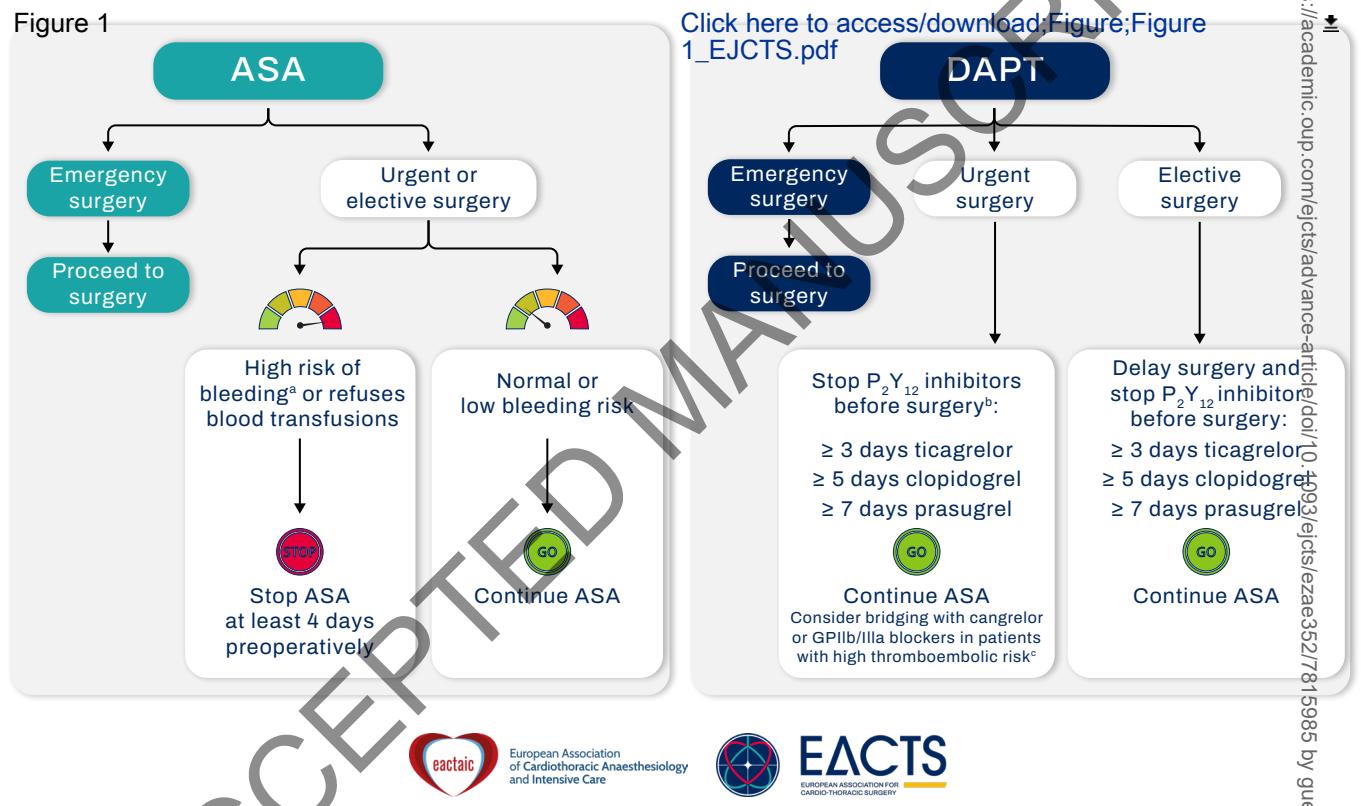
*Figure: Flowchart providing evidence-based guidelines for perioperative management of ASA and DAPT, balancing bleeding and thromboembolic risks*
**The following details perioperative interruption and resumption guidelines:**
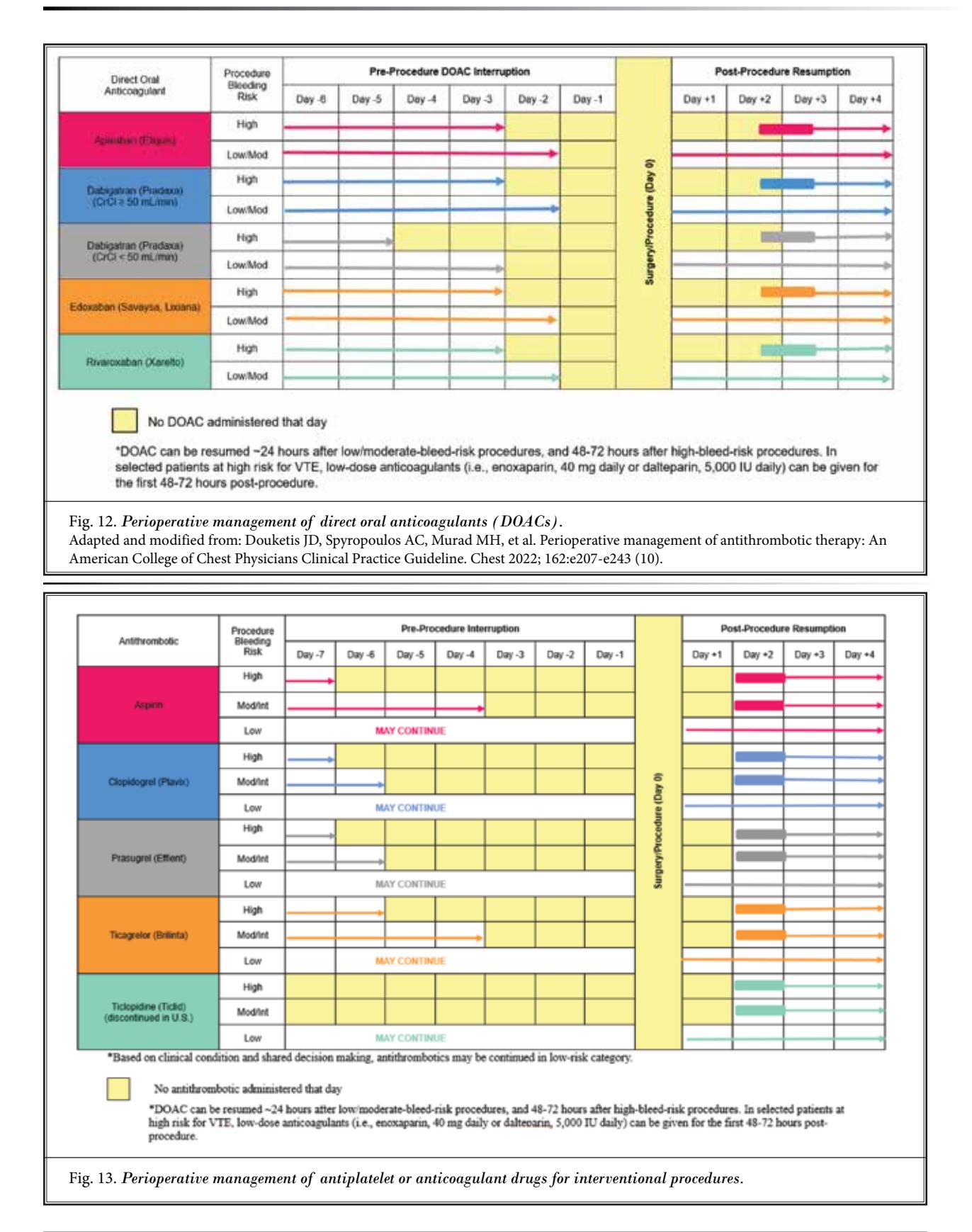
*Figure: Clinical guidelines for perioperative interruption and resumption of DOACs and antiplatelet medications based on procedure-specific bleeding risks*
**The following outlines the standardized perioperative management timeline:**
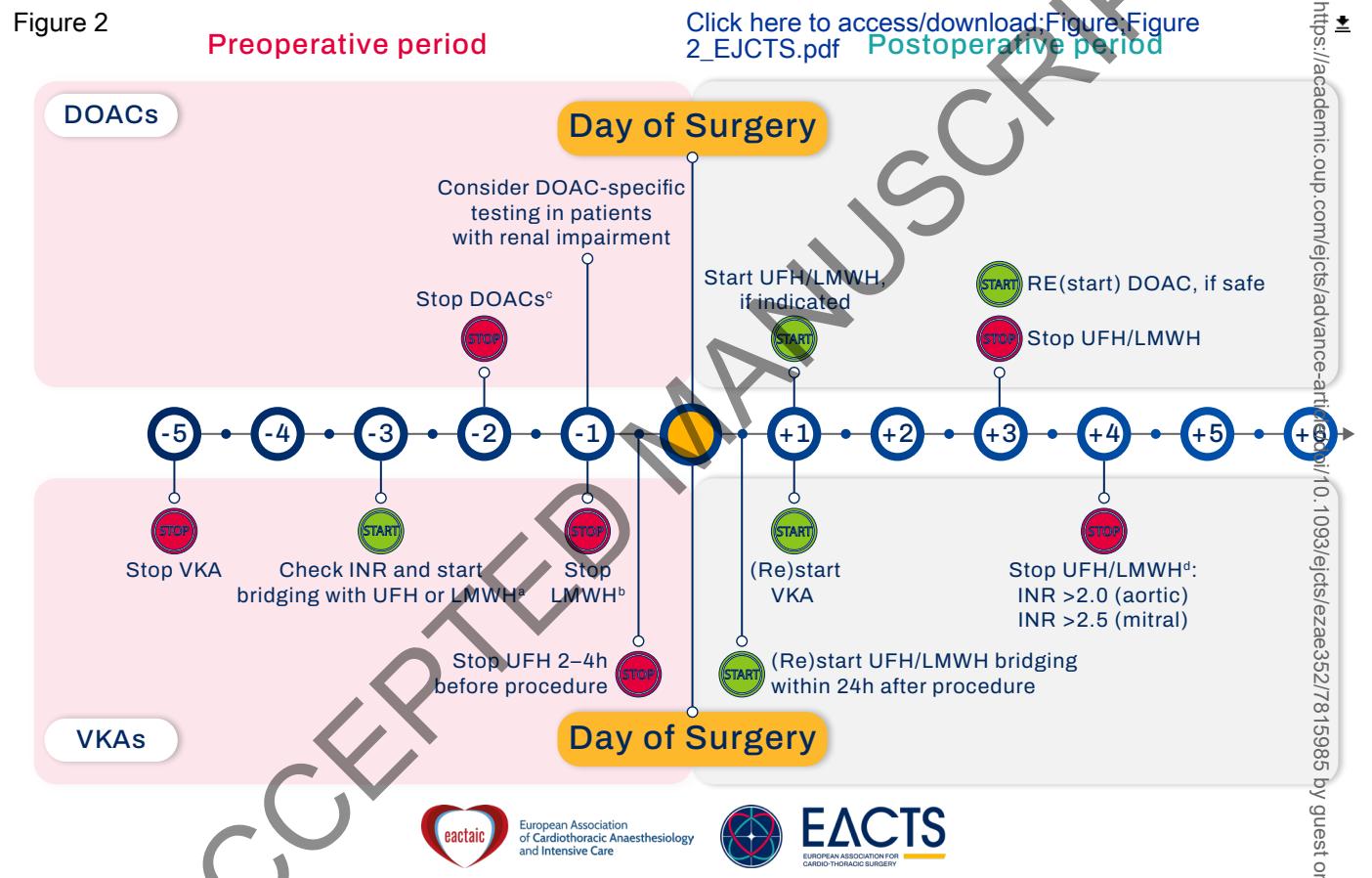
*Figure: Standardized perioperative management timeline for DOACs and VKAs, detailing specific cessation, bridging, and restarting points relative to the day of surgery*
---
### Thrombotic Risk Stratification and Bridging Indications
| Risk Category | Clinical Scenarios | Bridging Recommendation |
|---------------|-------------------|------------------------|
| **Low-moderate risk** | AFib without recent stroke, well-deployed stents >12 months, low-risk PCI | No bridging needed; continue or briefly interrupt antiplatelet therapy |
| **High risk** | Mechanical heart valves, recent (<12 weeks) stroke/TIA, recent stent thrombosis | May consider bridging with careful risk-benefit assessment |
| **Very high risk** | Recent stent placement (<30 days), left ventricular assist device, recent acute coronary syndrome | GP IIb/IIIa inhibitors (tirofiban, eptifibatide) or cangrelor preferred over LMWH [8] |
**Bridging protocol when indicated** [8]:
- Stop antiplatelet drugs **5-7 days** before planned surgery
- Start bridging therapy (GP IIb/IIIa inhibitors preferred over LMWH in very high-risk)
- Last dose **4-12 hours** before surgery (depending on agent)
- Restart postoperatively once surgical hemostasis is ensured
---
### Guideline Recommendations
**2024 EACTS/EACTAIC Guidelines on Patient Blood Management** [1][5]:
- Preoperative bridging of anticoagulants with LMWH or UFH is **not recommended** for most patients undergoing cardiac surgery
- The increased bleeding risk outweighs any potential thrombotic benefit
**2022 ACCP Clinical Practice Guideline** [9]:
- Addresses 43 PICO questions on perioperative management of antithrombotic therapy
- Recommends individualized risk stratification for bridging decisions
**ASIPP 2024 Guidelines** [6]:
- For low and moderate-risk interventional procedures, recommends **against cessation** of anticoagulant or antiplatelet therapy
- Emphasizes risk stratification by procedure type (low, moderate, high risk)
---
### Conclusion
The preponderance of clinical trial evidence demonstrates that LMWH bridging in DAPT patients increases major bleeding without reducing thrombotic events in most perioperative settings. Bridging should be reserved for very high thrombotic risk patients, where GP IIb/IIIa inhibitors or cangrelor may be preferred over LMWH. For the majority of patients, temporary interruption of antiplatelet therapy without bridging, or continuation through low-risk procedures, represents the evidence-based standard of care.
---
*This analysis is based on retrieved evidence from clinical trials and guidelines. Clinical decisions should consider individual patient thrombotic and bleeding risk profiles, and verify against current institutional protocols.*